Perspectives on Training Strategy in Life Sciences
Training in the medical device, biotech, and pharmaceutical industries carries a weight that other sectors don’t face. When training falls short, the consequences reach patients. KDG Life Science publishes research, frameworks, and field-tested guidance for organizations that treat training as a strategic function, not a pre-launch task.
Medical Device Development Lifecycle: Training Integration Framework
Overview
Provided is a phase-by-phase framework for embedding training strategy across the full medical device development lifecycle, from discovery through post-market support. This document maps seven development phases against the training considerations, stakeholder requirements, and deliverables that drive regulatory success and commercial adoption.
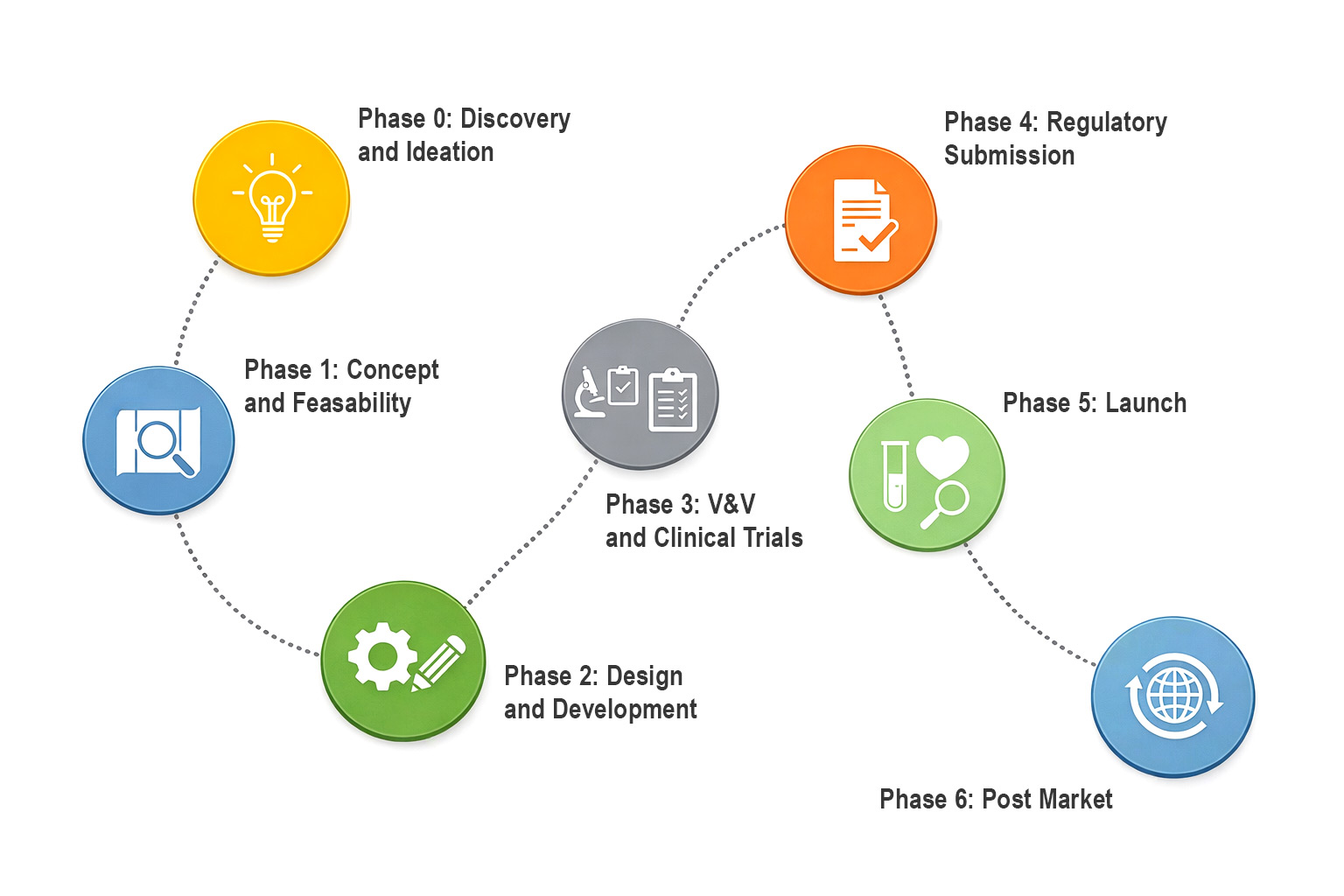
What’s inside:
Who it’s for:
Unlock the Key to a Successful Medical Device Launch
Overview
Bringing a medical device to market is more than just innovation, it’s about ensuring every stakeholder, from sales teams to physicians, has the confidence and knowledge to use it effectively.
This eBook delivers practical strategies to design impactful training, remove adoption barriers, and drive measurable results, turning education into a strategic advantage for your launch.

What’s inside:
Who it’s for:
About the Author
Lynette Kramer is a learning design expert and the founder of KDG Life Science. With decades of experience in medical device training, she has helped global companies create training programs that drive adoption, compliance, and sales success.

